The Jarrard Lab Research is Focused on Understudied Aspects of Epigenetic Tumor Biology.
Targeting histone-modifying enzymes to treat cancer
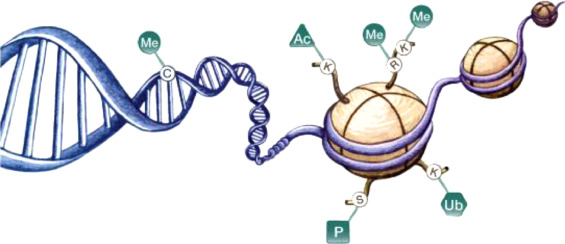 A recent aspect of our research addresses the role of histone-modifying enzymes, proteins that modify histone tails, during prostate cancer progression. Androgen deprivation therapy (ADT) is the standard treatment for advanced hormone-sensitive prostate cancer (HSPC), leading to initial tumor regression, but eventually, patients become resistant to ADT and transition to castration-resistant prostate cancer (CRPC). Both genomic and epigenomic alterations have been strongly implicated in the development of this disease.
A recent aspect of our research addresses the role of histone-modifying enzymes, proteins that modify histone tails, during prostate cancer progression. Androgen deprivation therapy (ADT) is the standard treatment for advanced hormone-sensitive prostate cancer (HSPC), leading to initial tumor regression, but eventually, patients become resistant to ADT and transition to castration-resistant prostate cancer (CRPC). Both genomic and epigenomic alterations have been strongly implicated in the development of this disease.
We showed that histone-modifying enzymes (HMEs) contribute to the survival of tumor cells during ADT. These changes are pivotal in the subsequent development of CRPC (Br J Cancer 2021, BMC Cancer 2017). Recent work has found that genetic mutations in chromatin remodelers may influence the expression and activity of HMEs during the development of CRPC (in review). These enzymes are attractive cancer targets that have the potential to provide novel personalized medicine approaches to prostate cancer therapy.
This is an accordion element with a series of buttons that open and close related content panels.
Why prostate cancer develops in men and how to prevent it
Why prostate cancer develops in men and how to prevent it
Our laboratory was the first to identify an epigenetic field of susceptibility involving an important oncogene (IGF2) in the histologically normal peripheral prostate of men with cancer (Clin Epigenetics. 2019, Prostate 2011). We subsequently found that this epigenetic defect occurs with aging and was more extensive in men with prostate cancer than in men who did not have the disease (Cancer Res. 2008). Engineering this epigenetic defect in IGF2 in mice leads to the development of prostate cancer (Cancer Res. 2017).
Further investigation into this field of cancer susceptibility using DNA methylation microarrays demonstrates a methylation ‘fingerprint’ in non-tumor prostate tissue that identifies men with prostate cancer (J Urol. 2014, Neoplasia 2013). Its detection in urine may be a more sensitive approach since it does not require the presence of cancer cells (Am J Clin Exp Urol. 2021). This work was the first to indicate that epigenetic changes in normal prostate tissue can be used to detect the disease from histologically negative biopsies, a discovery that has been used subsequently in the development of commercial markers.
This work was awarded the Wisconsin Alumni Research Innovation award for 2017 (out of 400 patents). Targeting the mechanism underlying this field defect is an ongoing focus (Clin Epigenetic. 2020) of our translational research work seeking to prevent or reverse these epigenetic changes. These findings present promising sources for prostate cancer screening as it is non-invasive and may be used to reduce the need for repeat biopsy, associated with unnecessary costs and complications.
Tumor response to androgen deprivation therapy exposes therapeutic susceptibilities
Tumor response to androgen deprivation therapy exposes therapeutic susceptibilities
Other research interests encompass improving the outcomes of patients with high-risk prostate cancer. Dr. Jarrard was the urology principle investigator on the ECOG 3805 CHAARTED trial that demonstrated metastatic HSPC patients treated with ADT and docetaxel led to a remarkable 14-month improvement in survival (N Engl J Med 2015). Research in the Jarrard Lab has sought to improve responses to ADT by studying synthetic lethality. We were the first to define a novel cellular response of senescence that occurs in response to ADT (PloS One. 2017, Prostate. 2013).
A Department of Defense grant led by our team screened and identified drugs that synergistically improve outcomes when combined with ADT. This work led to the demonstration that in 87,000 veterans, taking metformin at the time of initiating ADT for advanced prostate cancer improved cancer-specific and overall survival (J Urol. 2018). Metformin targets metabolic susceptibilities that ADT induces in the tumor (Mol Cancer Ther. 2020). Further investigation of this phenotype reveals other FDA-approved synergistic agents that have minimal toxicity (Prostate. 2022).
This work provides a rationale for the combined use of ADT with other agents in the treatment of metastatic HSPC – now a standard of care.
Clinical Research
Improving the clinical care of high-risk prostate cancer patients
Our clinical research seeks to improve the outcomes for high-risk prostate cancer by utilizing new imaging approaches and combination therapy. We have ongoing trials examining the use of chemohormonal therapy (funded through the Department of Defense and the UW Prostate Cancer SPORE) before surgery to remove the prostate.
Details about our clinical trials are below.
This is an accordion element with a series of buttons that open and close related content panels.
Can an anticancer vaccine improve outcomes when combined with surgery?
Can an anticancer vaccine improve outcomes when combined with surgery?
In ‘UW21015: a Phase I/II Trial of Androgen Deprivation, with or without pTVG-AR, and with or without Nivolumab, in Patients with Newly Diagnosed, High-Risk Prostate Cancer’, we are testing whether an anticancer vaccine can improve outcomes when combined with surgery.
Preclinical studies have demonstrated that androgen deprivation, sequenced with a DNA vaccine against the androgen receptor, can elicit prostate tumor-infiltrating lymphocytes and anti-tumor responses. Other studies have demonstrated that T-cell activation with vaccination leads to upregulation of PD-1 on CD8+ T cells, and that blockade of PD-1 at the time of vaccination leads to greater anti-tumor responses. The study aims to test pathological complete responses or minimal residual disease (MRD) that can occur in patients with prostate cancer treated with ADT and pTVG-AR, alone or in combination with nivolumab, before definitive surgery to remove the prostate.
If you have interest in this trial, contact our study coordinator:
Mary Jane Brennan, RN, BSN, at mjs@medicine.wisc.edu, Tel: (608)263-7107
Novel PSMA-PET imaging agent provides a new way of looking at prostate cancer
A novel PSMA-PET imaging agent offers a new way of looking at the disease before treatment as well as treatment responses
A current trial examines a radiotracer called Pylarify® (18F-DCFPyL) to determine if using this novel imaging agent in PET Scans improves cancer detection in low-risk prostate cancer patients compared to previously used diagnostic methods.
Pylarify, the radiotracer under study, binds to PSMA – a protein found mainly on prostate cancer cells – indicating precise cancer location and any spreading beyond the prostate. UW researchers believe that Pylarify will improve the effectiveness of cancer diagnosis over previous radiotracers because it has demonstrated a high tumor-to-background ratio and a high tumor-specific uptake when being developed, allowing for a greater chance of cancer detection. UW-Madison is one of the first and only research sites approved to study this new PET scan imaging agent in a cohort of low-risk prostate cancer patients.
The team has been recruiting study participants for roughly six months. Patients who qualify to participate in this study have prostate cancer requiring “active surveillance” (observation and testing) rather than active treatment, as the probability of their cancer spreading is low. Current standard-of-care procedures to follow their cancer progression include pelvic MRI and ultrasound-guided prostate biopsy. An MRI identifies cancerous lesions in the prostate and specific regions of the prostate that require biopsy in addition to the standard tested areas. However, MRI misses some cancers as it relies on differences in density and blood flow that may not be detectable in all tumors. These discrepancies and inaccuracies with MRI alone led the research team to examine PET scans using Pylarify as a new diagnostic option for these active surveillance patients.
If UW Researchers demonstrate that a Pylarify PET scan is a more sensitive and accurate method for cancer detection in low-risk patients as compared to standard-of-care imaging, a broader patient population will have access to a more powerful diagnostic tool that would lessen the chance of cancer being missed or spreading – ultimately allowing patients and their urologists to make better treatment decisions.
We are still recruiting participants for this study and have experienced a high level of interest – including from patients outside Wisconsin. If you would like to learn more, have questions, or are interested in joining the study, contact our study coordinator:
Abigail (Abby) Wiedmer, BS (Clinical research coordinator) at wiedmer@urology.wisc.edu, Tel: (608)265-9172
Pomegranate to improve prostate cancer active surveillance
Pomegranate to improve prostate cancer active surveillance
On the other end of the cancer spectrum, we have been employing molecular approaches to better inform clinical trials looking at the prevention of prostate cancer (Eur J Cancer Prev 2016; Am J Clin Exp Urol 2016). Active surveillance patients with a histologic diagnosis of organ-confined, low-grade prostate cancer with prostate tissue available for biomarker analysis were randomized to daily placebo or pomegranate extract capsules for a duration of 1-year.
Upon conclusion of the treatment period, collection of prostate tissue via biopsy for comparative quantitative immunohistochemical analysis was completed. This study demonstrates an important advancement in our understanding of the ability of pomegranate extract supplementation to penetrate prostate tissue and modulate oxidative stress and androgen signaling. This randomized multi-center study represents the first to specifically investigate pomegranate supplementation in the rapidly growing active surveillance population.
Jarrard Lab
Recent Publications
Perform a PubMed search for more of Dr. Jarrard’s publications
